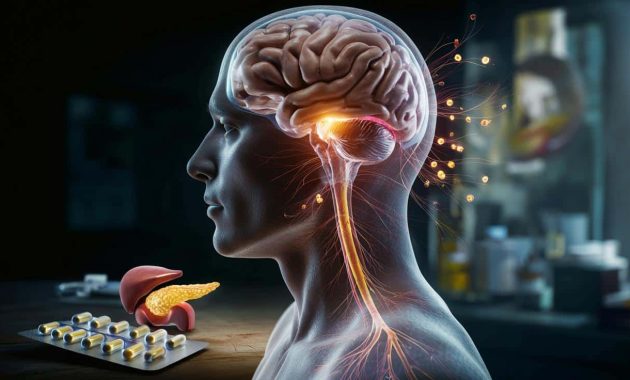
Now, more than sixty years after its arrival, researchers say this familiar drug may also be working in a far less obvious place: deep in the brain, where it appears to tweak circuits that control blood sugar, appetite and energy use.
A veteran diabetes drug back in the spotlight
Metformin is one of the most prescribed medicines on the planet. Since the 1960s, it has been the go‑to treatment for people with type 2 diabetes, helping millions bring their blood sugar under control at relatively low cost.
Doctors have long described its action in fairly simple terms. The drug improves the body’s sensitivity to insulin. It reduces the liver’s production of glucose. It helps muscles use sugar more efficiently. For decades, that story was enough to justify its routine use.
Yet a strange pattern kept emerging in clinical studies. People on metformin sometimes lived longer. Some appeared to have a lower risk of certain cancers. Others showed better cognitive performance than expected for their age and health status. These extra benefits looked real, but the mechanisms stayed fuzzy.
Metformin worked so well in practice that medicine accepted a partial explanation and moved on, leaving deeper questions unanswered.
That unfinished story has now gained a striking new chapter, and it shifts attention from the pancreas and liver to the brain itself.
The hypothalamus steps onto centre stage
A team at the Baylor College of Medicine in Texas, led by Professor Yong Xu, has been probing how metformin behaves in animal models. Their work, published in the journal Science Advances, suggests the drug acts directly on a brain region called the hypothalamus.
The hypothalamus is tiny but crucial. It sits at the base of the brain and acts as a command hub for basic bodily functions: hunger, thirst, body temperature, hormone release and, importantly, glucose metabolism.
In experiments on mice, Xu’s group showed that metformin affects a specific signalling pathway inside certain hypothalamic neurons. The key player is a protein known as Rap1, which helps regulate how cells respond to external signals.
➡️ Goodbye Footprint Marks on Sandals: The Simple Trick That Makes Them Look Brand New
➡️ Once deforestation halted, regional rainfall cycles began stabilizing
➡️ The 6 things you feel when you’re letting your life pass you by, according to psychologists
➡️ How cats show affection: when they do this, you are their favourite human
➡️ Why more and more gardeners are turning to lasagna gardening at the end of winter
➡️ How to use the new DLSS 4.5 with any Nvidia GeForce RTX graphics card?
➡️ Hang it by the shower: the clever bathroom hack that eliminates moisture and keeps your space fresh
By modulating the Rap1 pathway in discrete neurons, metformin appears to strengthen the body’s insulin response and curb the liver’s glucose output.
When the researchers disabled Rap1 in those neurons, metformin’s impact on blood sugar control changed dramatically. This pointed to the brain — not just the liver, muscles or fat tissue — as a central target of the drug.
From peripheral agent to brain–body coordinator
These findings tie together a broader shift in metabolic science. Over the last decade, evidence has accumulated that type 2 diabetes and obesity are not purely “peripheral” diseases. The central nervous system, especially the hypothalamus, plays a pivotal role in how the body manages energy and sugar.
Metformin now looks less like a simple glucose-lowering agent and more like a coordinator between brain circuits and metabolic organs. That perspective helps explain why its benefits sometimes extend beyond blood sugar readings.
- Improved communication between brain and liver may reduce excessive glucose production.
- Altered hypothalamic signalling can affect appetite and food choices.
- Better metabolic control in the brain might impact memory, mood and cognitive ageing.
What this means for future metabolic treatments
If the brain is a genuine primary target, drug development strategies could start to look different. Instead of focusing almost entirely on the pancreas, liver or gut, pharmaceutical research may pay closer attention to neurons that sense nutrients and hormones.
In the Baylor experiments, metformin’s action in the hypothalamus did more than adjust glucose. It also influenced how much the mice ate and how much energy they burned at rest, two critical levers for body weight.
Targeting brain circuits involved in energy balance could allow future drugs to address diabetes and obesity together, rather than as separate problems.
This idea has parallels with newer injectable drugs such as GLP‑1 receptor agonists, originally created for diabetes but now widely used for weight management. Those drugs also act partly on brain regions that regulate appetite. Metformin brings a different mechanism, centred on Rap1 and hypothalamic neurons, yet the concept is similar: treat metabolism by speaking directly to the brain.
Key points from the new research
| Aspect | What the study suggests |
|---|---|
| Primary site of action | Metformin influences neurons in the hypothalamus, not only liver and muscle cells. |
| Signalling pathway | The Rap1 protein in specific brain cells is central to its effect on insulin signalling. |
| Metabolic outcomes | Improved insulin sensitivity and reduced liver glucose production in mice. |
| Behavioural effects | Changes in food intake and energy expenditure, with implications for obesity. |
Why an old molecule can still surprise scientists
Metformin originates from a plant, French lilac, used in European folk remedies for centuries. The drug itself entered modern practice in the mid‑20th century. Because it is off‑patent, cheap and ubiquitous, it does not attract the same commercial push for research as newer, branded treatments.
Yet its sheer popularity and the hints of unexpected benefits keep drawing attention from academic laboratories. The new brain‑centred findings highlight how even well‑worn drugs can hold uncharted mechanisms.
For patients, this does not change how metformin is prescribed today. It remains a first‑line treatment for type 2 diabetes, typically used alongside lifestyle measures and, if needed, additional medications.
The immediate takeaway is not a new prescription, but a deeper appreciation of how intertwined brain health and metabolic health can be.
What this could mean for brain health and ageing
Several observational studies have linked metformin use with slower cognitive decline in some people with diabetes. Those studies have limits, and they do not prove cause and effect, yet they sparked interest in the drug as a potential ally for the ageing brain.
The hypothalamic mechanism described by the Baylor team offers one possible route by which a metabolic drug might influence brain function more broadly. Better control of blood sugar and insulin can shield tiny blood vessels in the brain, lowering the risk of small strokes and other damage. Stable energy supply may also support neurons that are particularly sensitive to glucose swings.
Researchers are now asking whether adjusting Rap1 and related pathways could help in conditions that sit at the crossroads of metabolism and neurology, such as Alzheimer’s disease, vascular dementia or depression linked with obesity.
Some terms worth unpacking
For anyone trying to follow this research, a few technical terms come up repeatedly:
- Type 2 diabetes: A chronic condition where the body’s cells do not respond properly to insulin, leading to raised blood sugar levels over time.
- Insulin resistance: When cells in muscles, fat and liver do not react well to insulin, forcing the pancreas to produce more of it to achieve the same effect.
- Hypothalamus: A small brain region controlling hunger, thirst, sleep, hormones and many automatic bodily processes.
- Rap1 protein: A molecular switch inside cells that helps process signals controlling growth, energy use and responses to hormones.
Real‑life questions for patients and doctors
People living with type 2 diabetes often juggle several concerns at once: blood sugar levels, weight, heart risk, and fears about future memory problems. A drug that nudges multiple systems at once can look appealing, but it also raises nuanced questions.
For instance, could combining metformin with newer brain‑active metabolic drugs deliver additive benefits, or would certain pathways overlap in unhelpful ways? Might people with pre‑diabetes one day receive low‑dose treatment aimed specifically at protecting brain circuits before full‑blown disease develops?
Clinicians also need to balance enthusiasm with caution. Metformin has known side effects, most commonly gastrointestinal discomfort. Rarely, it can trigger lactic acidosis, especially in people with severe kidney problems. Any move to use it for non‑diabetic conditions would require careful trials, not just extrapolation from current practice.
How this research could reshape everyday care
If future studies confirm the hypothalamic Rap1 mechanism in humans, the impact could reach beyond a single drug. Screening new compounds might start with questions such as: does this agent reach the brain? Which neurons does it influence? How does it alter the dialogue between brain, liver, fat and muscle?
Healthcare providers might also pay closer attention to cognitive symptoms in people with metabolic disease, treating brain fog, mood changes or mild memory issues not as side notes but as core features of the condition. Lifestyle measures already known to affect both brain and metabolic health — sleep quality, regular physical activity, resistance training, and diets rich in fibre and low in ultra‑processed food — could gain additional scientific backing when framed through this brain‑centred model.
For now, the new data on metformin’s brain effects act as a reminder that even a familiar pill can reshape thinking about how the body runs itself, from the first bite of food to the firing of deep, unseen neurons.
Originally posted 2026-03-12 04:25:19.